Biologic Therapies, Inc. Begins Operations with Groundbreaking New Medical Devices
It was announced today that a new medical device company, Biologic Therapies, Inc., has begun operations in Ocala, Florida.
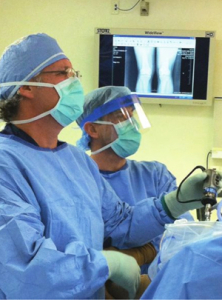
Ocala, FL, October 20, 2011 --(PR.com)-- Biologic Therapies specializes in the design and manufacturing of components for the biologics sector of the medical device industry. The company was formed as a joint venture between Scorpion Medical, Inc., a precision manufacturer of medical devices, and Monet Medical, Inc., a medical device development company owned and operated by Dr. R. Wade McKenna, an orthopedic surgeon, and Charles Bolles, a medical device distribution expert. Biologic Therapies was formed to provide proprietary, groundbreaking medical technologies that management anticipates will significantly enhance physicians’ use of Orthobiologic materials and surgical procedures for the treatment of orthopedic conditions.
By definition, Orthobiology is the inclusion of biology and biochemistry in the development of bone and soft tissue replacement materials for skeletal and tissue healing. Biologic Therapies intends to take this process to a new level by creating ‘Autologous Orthobiologic Therapy’. “We believe we can utilize the body’s own stem cells to increase the healing potential and provide quicker restoration of function within muscle, tendon, ligament, bone and cartilage,” says Dr. McKenna.
According to the 2010 Orthopedic Industry Annual Report, the field of Orthobiologics is among the fastest growing market segments in the orthopedic industry. Orthobiologic revenue reached $4 billion in 2010, which included sales of bone and soft tissue substitutes, allograft bone/bone proteins, stem cells, hyaluronic acid and more.
With over 30 years of success in the medical device industry with companies including DePuy / a Johnson & Johnson company, Encore Medical and Unified Strategies, Steve Bales is the President of Biologic Therapies. Bales states, “We are very pleased to formalize the partnership between Scorpion Medical and Monet Medical. This collaboration will combine the precision manufacturing capabilities of Scorpion Medical with Dr. McKenna’s extensive clinical experience and Mr. Bolles’ wealth of knowledge and contacts in the medical device industry. We believe that Dr. McKenna’s experience with stem cell therapy will enable Biologic Therapies to stay at the forefront of this new technology and react to the needs of surgeons supplying patients with the latest Orthobioligic systems available. Stem cell therapy is now being proven in a number of medical specialties and we expect to build a very successful pipeline of products to effectively meet the needs of this emerging market.”
Dr. McKenna’s autologous biologic therapy (bone marrow aspirate stem cells) treatments are designed to augment overall healing and improve surgical and non-surgical results. The treatments are for ligamentous injuries involving the knee, ankle and elbow. Treatments in the knee include ACL, MCL and LCL injuries. In the elbow, stem cells are used successfully to treat acute and chronic medial and lateral epicondylitis. Ligamentous injuries around the ankle are referred to as sprains but can often represent significant tears to the stabilizing structures of the ankle. Autologous biologic therapy is used around the ankle in chronic and acute sprains to shorten the recovery time and improve the quality of healing tissue.
In the shoulder, Dr. McKenna utilizes autologous stem cells and growth factor therapy to improve the outcomes of rotator cuff repairs. Rotator cuff injuries that require surgery due to their severity greatly benefit from the use of autologous biologic therapy. Recently, autologous biologic therapy was successfully used to address the problem of chondrolysis in the shoulder. Chondrolysis is a condition that most often leads to shoulder replacement surgery.
Perhaps the most exciting use of autologous biologic therapy involves the use of stem cells to promote healing and regeneration of cartilage and bone. Chondrogenesis, or the growth of cartilage, is the goal of treatment. With nearly 100 patients treated by Dr. McKenna, autologous biologic therapy shows great promise in addressing articular cartilage defects caused by arthritis or trauma as an augment to micro fracture surgery.
To date, the most widespread accepted current use involving autologous biologic therapy has been to improve outcomes and shorten recovery time in spinal fusion surgery. In many difficult fractures autologous stem cells have been used to improve the results of fracture healing and have been applied to non-unions with tremendous success. At Biologic Therapies, the goal is to increase the awareness of the enormous benefits associated with autologous biologic therapy in both surgical and non-surgical applications.
Many medical researchers believe that stem cell treatments have the potential to change the face of human disease and help alleviate suffering. The ability of stem cells to self-renew and give rise to subsequent generations with variable degrees of differentiation capacities offers significant potential for generation of tissues that can potentially replace diseased and damaged areas in the body with minimal risk of rejection and side effects.
Regenerative medicine, or simply put, the body using its own properties to heal itself, is currently considered by many as the future of medicine. Adult Mesenchymal Stem Cells are found in bone marrow and act to repair orthopedic problems. Management believes this venture positions Biologic Therapies as one of the pioneers in this new and exciting field.
As its first product offering, Biologic Therapies is actively working to bring to market a new, patented device for accessing a patient’s own stem cells through the safe and painless harvest of bone marrow. This breakthrough device is scheduled for market release during the first quarter of 2012. Management believes this device is unlike anything currently available and expects it could become the ‘gold standard’ for use in stem cell harvesting procedures.
Closely following the stem cell harvesting device will be a family of implant systems dedicated to fracture fixation, which will include the ability to introduce stem cells precisely at the fracture site. In addition, Biologic Therapies intends to create a strategic initiative to broaden the training and product support by offering an educational element to surgeons and healthcare professionals. The educational program will be dedicated to enhance the awareness and efficacy of this emerging technology as well as share Biologic Therapies’ surgical protocols designed to increase the success rate of Autologous Orthobiologic Therapies.
Acting as Biologic Therapies’ Chief Medical Director, Dr. McKenna has been intimately involved in this new medical science for nearly three years and has extensive clinical data to support the use of stem cell therapy. Dr McKenna is currently providing Autologous Orthobiologic Therapy to his patients and is very encouraged by the results. “Our results have been fantastic. I’m extremely encouraged. We have gone from ‘treating’ orthopedic related soft tissue injuries and bone fractures to ‘healing’ them with great success. Our work and clinical data has us much further along than most,” states Dr. McKenna.
Chuck Bolles, Vice President of Sales & Distribution at Biologic Therapies has extensive experience in the medical device distribution industry. Management believes his expertise will help to ensure the success of this new venture. Bolles states, “We have a responsibility to the medical community to continue our work in finding expanded usage for stem cell therapy. It truly is one of the fastest growing segments of medicine, which deserves our serious attention.”
Robert Stopanio, President of Scorpion Medical, Inc., states, “This is a very exciting time for Scorpion Medical. As the exclusive manufacturer for all Biologic Therapies devices, we believe this new venture puts Scorpion Medical at the forefront of a new wave of technology including not only orthopedics, but other surgical specialties as well. We look forward to manufacturing devices that will help doctors treat patients in more effective ways and that will result in shorter recovery times.”
For more information on stem cells and stem cell therapies visit the National Institutes of Health web site at http://stemcells.nih.gov/info/basics.
###
By definition, Orthobiology is the inclusion of biology and biochemistry in the development of bone and soft tissue replacement materials for skeletal and tissue healing. Biologic Therapies intends to take this process to a new level by creating ‘Autologous Orthobiologic Therapy’. “We believe we can utilize the body’s own stem cells to increase the healing potential and provide quicker restoration of function within muscle, tendon, ligament, bone and cartilage,” says Dr. McKenna.
According to the 2010 Orthopedic Industry Annual Report, the field of Orthobiologics is among the fastest growing market segments in the orthopedic industry. Orthobiologic revenue reached $4 billion in 2010, which included sales of bone and soft tissue substitutes, allograft bone/bone proteins, stem cells, hyaluronic acid and more.
With over 30 years of success in the medical device industry with companies including DePuy / a Johnson & Johnson company, Encore Medical and Unified Strategies, Steve Bales is the President of Biologic Therapies. Bales states, “We are very pleased to formalize the partnership between Scorpion Medical and Monet Medical. This collaboration will combine the precision manufacturing capabilities of Scorpion Medical with Dr. McKenna’s extensive clinical experience and Mr. Bolles’ wealth of knowledge and contacts in the medical device industry. We believe that Dr. McKenna’s experience with stem cell therapy will enable Biologic Therapies to stay at the forefront of this new technology and react to the needs of surgeons supplying patients with the latest Orthobioligic systems available. Stem cell therapy is now being proven in a number of medical specialties and we expect to build a very successful pipeline of products to effectively meet the needs of this emerging market.”
Dr. McKenna’s autologous biologic therapy (bone marrow aspirate stem cells) treatments are designed to augment overall healing and improve surgical and non-surgical results. The treatments are for ligamentous injuries involving the knee, ankle and elbow. Treatments in the knee include ACL, MCL and LCL injuries. In the elbow, stem cells are used successfully to treat acute and chronic medial and lateral epicondylitis. Ligamentous injuries around the ankle are referred to as sprains but can often represent significant tears to the stabilizing structures of the ankle. Autologous biologic therapy is used around the ankle in chronic and acute sprains to shorten the recovery time and improve the quality of healing tissue.
In the shoulder, Dr. McKenna utilizes autologous stem cells and growth factor therapy to improve the outcomes of rotator cuff repairs. Rotator cuff injuries that require surgery due to their severity greatly benefit from the use of autologous biologic therapy. Recently, autologous biologic therapy was successfully used to address the problem of chondrolysis in the shoulder. Chondrolysis is a condition that most often leads to shoulder replacement surgery.
Perhaps the most exciting use of autologous biologic therapy involves the use of stem cells to promote healing and regeneration of cartilage and bone. Chondrogenesis, or the growth of cartilage, is the goal of treatment. With nearly 100 patients treated by Dr. McKenna, autologous biologic therapy shows great promise in addressing articular cartilage defects caused by arthritis or trauma as an augment to micro fracture surgery.
To date, the most widespread accepted current use involving autologous biologic therapy has been to improve outcomes and shorten recovery time in spinal fusion surgery. In many difficult fractures autologous stem cells have been used to improve the results of fracture healing and have been applied to non-unions with tremendous success. At Biologic Therapies, the goal is to increase the awareness of the enormous benefits associated with autologous biologic therapy in both surgical and non-surgical applications.
Many medical researchers believe that stem cell treatments have the potential to change the face of human disease and help alleviate suffering. The ability of stem cells to self-renew and give rise to subsequent generations with variable degrees of differentiation capacities offers significant potential for generation of tissues that can potentially replace diseased and damaged areas in the body with minimal risk of rejection and side effects.
Regenerative medicine, or simply put, the body using its own properties to heal itself, is currently considered by many as the future of medicine. Adult Mesenchymal Stem Cells are found in bone marrow and act to repair orthopedic problems. Management believes this venture positions Biologic Therapies as one of the pioneers in this new and exciting field.
As its first product offering, Biologic Therapies is actively working to bring to market a new, patented device for accessing a patient’s own stem cells through the safe and painless harvest of bone marrow. This breakthrough device is scheduled for market release during the first quarter of 2012. Management believes this device is unlike anything currently available and expects it could become the ‘gold standard’ for use in stem cell harvesting procedures.
Closely following the stem cell harvesting device will be a family of implant systems dedicated to fracture fixation, which will include the ability to introduce stem cells precisely at the fracture site. In addition, Biologic Therapies intends to create a strategic initiative to broaden the training and product support by offering an educational element to surgeons and healthcare professionals. The educational program will be dedicated to enhance the awareness and efficacy of this emerging technology as well as share Biologic Therapies’ surgical protocols designed to increase the success rate of Autologous Orthobiologic Therapies.
Acting as Biologic Therapies’ Chief Medical Director, Dr. McKenna has been intimately involved in this new medical science for nearly three years and has extensive clinical data to support the use of stem cell therapy. Dr McKenna is currently providing Autologous Orthobiologic Therapy to his patients and is very encouraged by the results. “Our results have been fantastic. I’m extremely encouraged. We have gone from ‘treating’ orthopedic related soft tissue injuries and bone fractures to ‘healing’ them with great success. Our work and clinical data has us much further along than most,” states Dr. McKenna.
Chuck Bolles, Vice President of Sales & Distribution at Biologic Therapies has extensive experience in the medical device distribution industry. Management believes his expertise will help to ensure the success of this new venture. Bolles states, “We have a responsibility to the medical community to continue our work in finding expanded usage for stem cell therapy. It truly is one of the fastest growing segments of medicine, which deserves our serious attention.”
Robert Stopanio, President of Scorpion Medical, Inc., states, “This is a very exciting time for Scorpion Medical. As the exclusive manufacturer for all Biologic Therapies devices, we believe this new venture puts Scorpion Medical at the forefront of a new wave of technology including not only orthopedics, but other surgical specialties as well. We look forward to manufacturing devices that will help doctors treat patients in more effective ways and that will result in shorter recovery times.”
For more information on stem cells and stem cell therapies visit the National Institutes of Health web site at http://stemcells.nih.gov/info/basics.
###
Contact
Biologic Therapies, Inc.
Scott Reynolds
352-512-0778
www.biologictherapies.com
Scott Reynolds
352-512-0778
www.biologictherapies.com
Multimedia
Categories